Asthma is a disease of the airways, characterised by airflow obstruction that varies over time, with symptoms including an unproductive cough, chest tightness, breathlessness, and wheeze that is detected by chest auscultation. Symptoms are brought on by triggers such as cold weather, respiratory virus, exercise, allergens, and stress (British Thoracic Society (BTS) / Scottish Intercollegiate Guideline Network (SIGN), 2019).
When exposed to a trigger, the smooth muscle that surrounds the airway contracts (bronchoconstriction) and the inside mucosal lining becomes inflamed (Kudo et al, 2013). Treatment involves bronchodilator and anti-inflammatory drugs, which are mostly delivered straight to the lungs via an inhaler device (National Institute for Health and Care Excellence (NICE), 2017; BTS/SIGN, 2019; Global Initiative for Asthma (GINA), 2020).
When compared to oral medication, inhaled therapy works faster, requires lower doses, and has fewer side effects (Allen et al, 2003). Effective inhaled therapy requires knowledge and ability to use an inhaler device, and the patient should be shown how to use this and have their technique checked by a healthcare professional who is competent to do so. Treatment relies on evidence-based inhaled therapy, with both drug and device being prescribed. Diagnosis and management is largely undertaken in primary care, with complex cases supported by secondary care and ‘difficult asthma’ services (NICE, 2017; BTS/SIGN, 2019; GINA, 2020).
It should be stressed that while this paper focuses on drugs rather than inhaler devices, the subject of devices is large and complex, and its importance is no less significant. The aim of asthma management is control of the disease (Bateman et al, 2004), defined as:
- No daytime symptoms
- No night-time waking as a result of asthma
- No need for rescue medication
- No asthma attacks
- No limitations on activity, including exercise
- Normal lung function (forced expired volume in 1 second or peak expiratory flow rate >80% of predicted or best).
Medication for asthma can be broadly classified into two main headings: anti-inflammatories and bronchodilators. In recent years, clinical guidelines have increasingly emphasised the importance of inhaled corticosteroids, which can now be seen as first-line treatment choice in adults and older children (BTS/SIGN, 2017; GINA, 2020).
Anti-inflammatories
Inhaled corticosteroids
Asthma is an inflammatory condition, and failure to treat inflammation in the airways leads to increased symptoms, loss of control, and increased risk of asthma attack and death (Royal College of Physicians, 2014). Inhaled corticosteroids (ICS) are the mainstay of asthma therapy, and remain the safest and most effective way of treating asthma. ICS work by reducing the number of circulating eosinophils, and subsequently reduce airway inflammation. They have also been shown to reduce airway hyper-responsiveness to triggers such as allergens, exercise, cold air and chemical irritants. ICS also have a positive effect on B2 receptors of airway smooth muscle, which may positively impact bronchodilators, while tobacco smoke has been shown to reduce clinical effectiveness (BTS/SIGN, 2019).
When compared with bronchodilators, ICS tend to have a much slower onset of action, often taking days or weeks before the patient perceives any symptomatic benefit or an improvement in lung function. ICS are generally dose-responsive and clinical effectiveness can increase with moderate doses (Kankaanranta et al, 2004). They are generally safe, well-tolerated, and the most common side effects are sore throat, hoarse voice, or oral candida. The risk of side effects can be reduced by asking the patient to rinse their mouth after use, or through the use of a spacer (NICE, 2020a). Beclometasone is the most common of the five ICS available for prescribing in England (Figure 1). Qvar has extra fine particles, and is twice as potent as Clenil (NICE, 2020b).
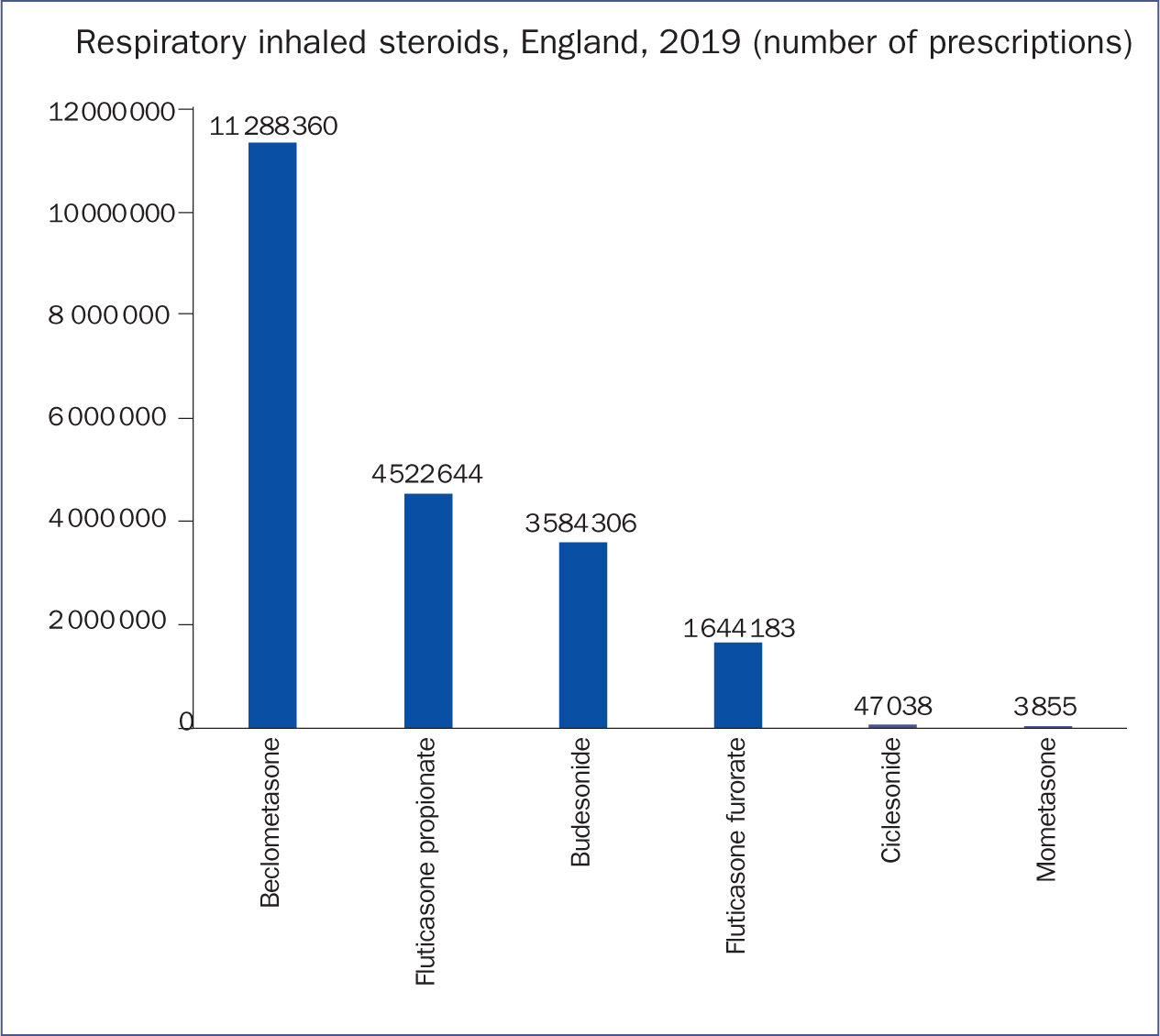 Figure 1. Inhaled steroids prescribed in England, 2019
Figure 1. Inhaled steroids prescribed in England, 2019
Oral treatments
While the intention of this article is to review inhaled therapies, it is also worth considering oral therapies for asthma.
Oral steroids
Used during an asthma worsening or asthma attack, oral steroids can be clinically effective at enabling symptom and lung function recovery (Rowe et al, 2007). Prednisolone 40 mg to 60 mg per day for a period of 5–7 days is usually prescribed, with a second weeks course may sometimes be needed if severe symptoms persist. Some patients with more severe asthma may be given a standby course of prednisolone to keep at home, as part of their individualised self management plan.
Patients should be reminded to book an urgent appointment for review if they commence their rescue oral steroids. However, they are not without side effects, which can include gastrointestinal discomfort, headache; impaired healing, osteoporosis, mood alteration and anxiety, weight gain. Because of this, regular oral steroid use should be strictly limited to specialist respiratory care only (BTS/SIGN, 2019).
Leukotriene receptor antagonists
Cysteinyl leukotrienes are inflammatory mediators released from mast cells and eosinophills that contribute to the inflammation and bronchoconstriction observed in asthma (Sarau et al, 1999). Leukoteriene receptor antagonists (LTRA) bind to the CysLT1 receptor found in the airway smooth muscle and decrease eosinophils within the airway and the blood, leading to a reduction in both airway inflammation and bronchoconstriction (Medicines.org.uk, 2016).
Used in conjunction with ICS, LTRA therapy has been shown to improve symptom control. A systematic review by Chauhan and Ducharme (2014) showed that the addition of LTRA to ICS therapy, was slightly less clinically effective than the addition of Long Acting B2 Agonist (LABA) to ICS. There is conflicting advice between the two UK guidelines regarding the addition of either LTRA or LABA (NICE, 2017; BTS/SIGN 2019), and the choice of which to prescribe may be down to clinician experience or patient preference.
Specialist treatments: anti IgE
For severe or difficult asthma, a relatively new treatment exists for initiation by specialist care only. Monoclonal antibody (MAB) injections are given by intramuscular injection once a month to control asthma, and can reduce the number or courses of oral steroids.
Specialist asthma services can initiate and monitor injectable therapy for severe, persistent allergic asthma (NICE, 2013) and for eosinophilic asthma. These patients are usually already on long-term oral steroids. MAB binds to Immunoglobulin E (IgE), thus inhibiting the amount of free IgE able to set off the inflammatory cascade (Medicines.org.uk, 2019a). Other MABs target interleukin-5 (IL-5) to reduce the production of eosinophils (Medicines.org.uk, 2019b).
Bronchodilators
Beta 2 agonists — short-acting
Beta 2 agonists stimulate the beta-two adrenoreceptors in the smooth muscle surrounding the airway, which results in bronchodilation in people with airway obstruction (Bourke and Burns, 2011). These can be sub-categorised into short-acting B2 agonists (SABA) and long-acting B2 agonists (LABA).
The speed of onset of SABA is 3 minutes (Seberova and Anderson, 2000), making them ideal for use as a ‘reliever’ of asthma symptoms, and are prescribed ‘as required’. Two SABAs are available, salbutamol and terbutaline, which are both highly dose-responsive, meaning that multiple doses can be given with increasing clinical benefit. Both also have bronchodilation effects lasting approximately 4 hours, hence the designation ‘short-acting.’ Inhaled SABA is also the treatment of choice during an asthma attack, where multiple doses can be given ‘back-to-back’ either through a pressurised metered dose inhaler (MDI) and large volume spacer, or via a nebuliser driven via air or, preferably, oxygen.
Beta 2 agonists — long-acting
LABA have a duration of action lasting either 12 (Rabe et al, 1993) or 24 hours (Lötvall et al, 2012). Most LABA have a relatively slow onset of action of approximately 20 minutes; however, formoterol acts as fast as SABA, providing bronchodilation and symptom relief within three minutes that lasts for 12 hours (Seberova and Anderson, 2000). Formoterol is also unique among LABAs in that it is dose-responsive and can be given multiple times in a day (Palmquivst et al, 1997). Conversely, salmeterol has a much lower therapeutic window, and can only be administered at a fixed dose (Bennett and Tattersfield, 1997).
Side effects of LABA and SABA therapy include peripheral tremor, tachycardia, anxiety, headache and muscle cramps, which are usually rare and limited to the duration of action (Bourke and Burns, 2011). Those patients who are sensitive to one B2 agonist, sometimes experience no side effects with another.
A third LABA is licensed for asthma, Vilanterol, which is available in combination with the ICS fluticasone furoate (Relvar Ellipta 184 micrograms/22 micrograms inhalation powder, pre-dispensed). Summary of product characteristics can be found on Medicines.org.uk (2019c).
Muscarinic antagonists — short-acting
Muscarinic antagonists are less effective in asthma than SABA and LABA (Bourke and Burns, 2011). They compete with acetylcholine at the muscarinic cholinergic receptors within the parasympathetic nervous system, which inhibits bronchoconstriction and allows the airways to dilate. Common side effects include cough, throat irritation, headache, dizziness, and cramps (Simonsen et al, 2006).
There is only one short-acting muscarinic antagonis (SAMA): ipratropium, more commonly known by its brand name of Atrovent. The speed of onset is approximately 15 minutes, and the duration of action approximately 4–6 hours. (Medicines.org.uk, 2018). It is not recommended in the routine treatment of asthma, but can be given in an asthma attack when it is nebulised conjointly with salbutamol (BTS/SIGN, 2019).
Muscarinic antagonists — long-acting
Although there are four long-acting muscarinic antagonists (LAMA), only one is available for asthma: tiotropium via the Respimat® device. This is licensed for asthma when ICS and LABA therapy have not been successful in maintaining control. Although they may be used conjointly with B2 agonists, they should not be used with another muscarinic antagonist (Medicines.org.uk, 2019d).
Devices
When prescribing inhaled therapy, both the drug and the device must be prescribed, preferably by brand (BTS/SIGN, 2019). Without this specification, the prescriber cannot be certain that the patient will receive the correct device. The patient must be shown how to use the device and their technique assessed to ensure they are using it correctly before the medication is issued (NICE, 2017; BTS/SIGN, 2019). In this way, the prescribing of the specific device, is as important as prescribing the route, dose, or frequency of a drug.
The subject of inhaler devices can appear complex and a speciality in its own right. Although this article does not aim to address the subject of inhaler technique in full, it may help to provide a basic introduction. More and more generic inhalers have flooded the marketplace in recent years, and there are now 16 inhaler devices that are licensed for asthma (Booth, 2020). Inhaler devices can be classified into two main groups: dry powder inhalers (DPI) and aerosol inhalers (also known as metered dose inhalers (MDIs)). These two groups of inhalers require different techniques, leading to a suggestion that it may be better for a patient to have either one or the other, but not both. One of the key differences relates to inspiratory flow rate; in other words, the speed or force at which the patient can inhale.
MDIs require a very low inspiratory flow rate (Sanchis et al, 2013), while DPIs require a high inspiratory flow rate (Haidl et al, 2016). A common error with the MDI is inhaling too rapidly, causing more drug to be deposited in the oropharynx, significantly reducing its effectiveness (Giraud and Roche, 2002). A potential difficulty with DPIs is if the patient's inspiratory flow rate is too low, causing the powder to remain inside the device, thus nullifying its effectiveness (Van Der Palen, 2003; Haidl et al, 2016).
When selecting a device for an individual patient, the inspiratory flow rate of the patient must be considered, along with the patient's ability to use the device, and their own personal preference. Inspiratory flow rate can be measured using the InCheck Dial®, or via an inspiratory flow training whistle, many of which are available from the manufacturer of the device (Sanders, 2017).
Aerosol devices
These comprise of a canister of compressed gas containing the drug and (in most cases) a propellant. Once the device is activated, the propellant is released, and the user inhales the drug.
Metered dose inhaler – self-actuated
Probably the most recognised inhaler device is the MDI. Compressing the cannister activates the device and the drug is expelled through a venturi and out through the mouthpiece. Coordination between activation and inhalation and ensuring the inspiratory flow rate is low is essential if its use is to be effective (Al-Showair et al, 2007; Haidl et al, 2016). With a perfect technique, only 10% of the drug will be deposited in the lungs. To improve lung deposition, a spacer, also known as a valved holding chamber, may be used, and is the preferred method of drug delivery in young children (BTS/SIGN, 2019). Spacers also enable the patient to take the MDI without the need for coordinating activation and inhalation, but it is acknowledged that some patients prefer not to use spacers. Some MDIs have dose counters, but others do not, making it impossible to tell for certain if the device contains any further drug. By shaking the canister, it may be possible to detect the presence of fluid inside, which may just be propellant.
The Respimat®, is an inhaler that may be considered in its own right. Also known as a ‘soft mist’ inhaler, the technology utilised relies on a mechanical spring, rather than a propellant, to expel the drug. The resulting drug is dispersed in a mist or vapour that has smaller particles, and increases lung deposition (Dalby et al, 2004).
Metered dose inhalers – self-actuated
The Easybreathe®, Autohaler®, and K-haler® devices are MDIs that are automatically activated as the patient begins inhaling. This eliminates the need for coordinating activation and inhalation. Breath actuated inhalers do not have a dose counter, with the exception of the K-haler®, which has a dose counter on the front.
Dry powder inhalers
Although they require a higher inspiratory flow rate than aerosol inhalers, evidence shows that DPIs can still be used during an asthma attack (Selroos et al, 2009). Also, there is no need to coordinate activation with inhalation, and most have a dose counter.
Dry power inhalers – capsule
Capsule DPIs involve removing a capsule from a bottle or blister pack, and placing the capsule inside the device. The capsule is then pierced by pressing a button on the device, which sends a spike through the plastic capsule wall, priming the device for usage, and the powder is then inhaled as forcibly as possible, before the capsule is discarded. These require a degree of manual dexterity, and run the risk of the capsule being mistaken for a tablet and swallowed.
Dry powder inhalers — multi-dose
Most DPIs involve opening a cover, twisting the base, or pressing a button to load or prime the device. The powder can then be inhaled as forcibly as possible. A dose counter indicates how many doses are left and when empty, the device can be discarded.
Guidelines
National and international guidelines show that the aim of asthma management is to control symptoms (NICE, 2017; BTS/SIGN, 2019; GINA, 2019). Assessing control of asthma is an important element of prescribing, which is best undertaken using a structured asthma questionnaire such as the Asthma Control Test (Nathan et al, 2004) or the Royal College of Physicians Three Questions (Pinnock et al, 2012). Therapy should be increased to improve control and reduced when well-controlled, to establish the lowest controlling therapy. Asthma control should be assessed at every patient contact and before any consideration to change prescription, along with the patient's inhaler technique and adherence to their agreed treatment plan. Adherence to medication is an important consideration, as is patient preference (Scullion, 2020) and engagement with the patient is essential when discussing and selecting any therapy for asthma (NICE, 2017; BTS/SIGN 2019; GINA, 2019).
The following is based upon the BTS/SIGN (2019) asthma guidelines and consideration has been also made for the NICE guidance on LTRA therapy. Prescribing examples given below are for children, adults (DPI) and adults (MDI) and additional resources to aid prescribing of inhaled therapy are freely available online via Rightbreathe (NHS London Procurement Partnership, 2020) and the Primary Care Training Centre (Booth, 2020). Figure 2 provides three diagrammatic examples of typical pharmacological pathways through these guidelines. Historically, guidelines have dealt with bronchodilator therapy first. However, as asthma is an inflammatory condition, and as more modern guidelines stress the use of inhaled steroids as a first-line treatment choice, these have been considered first.
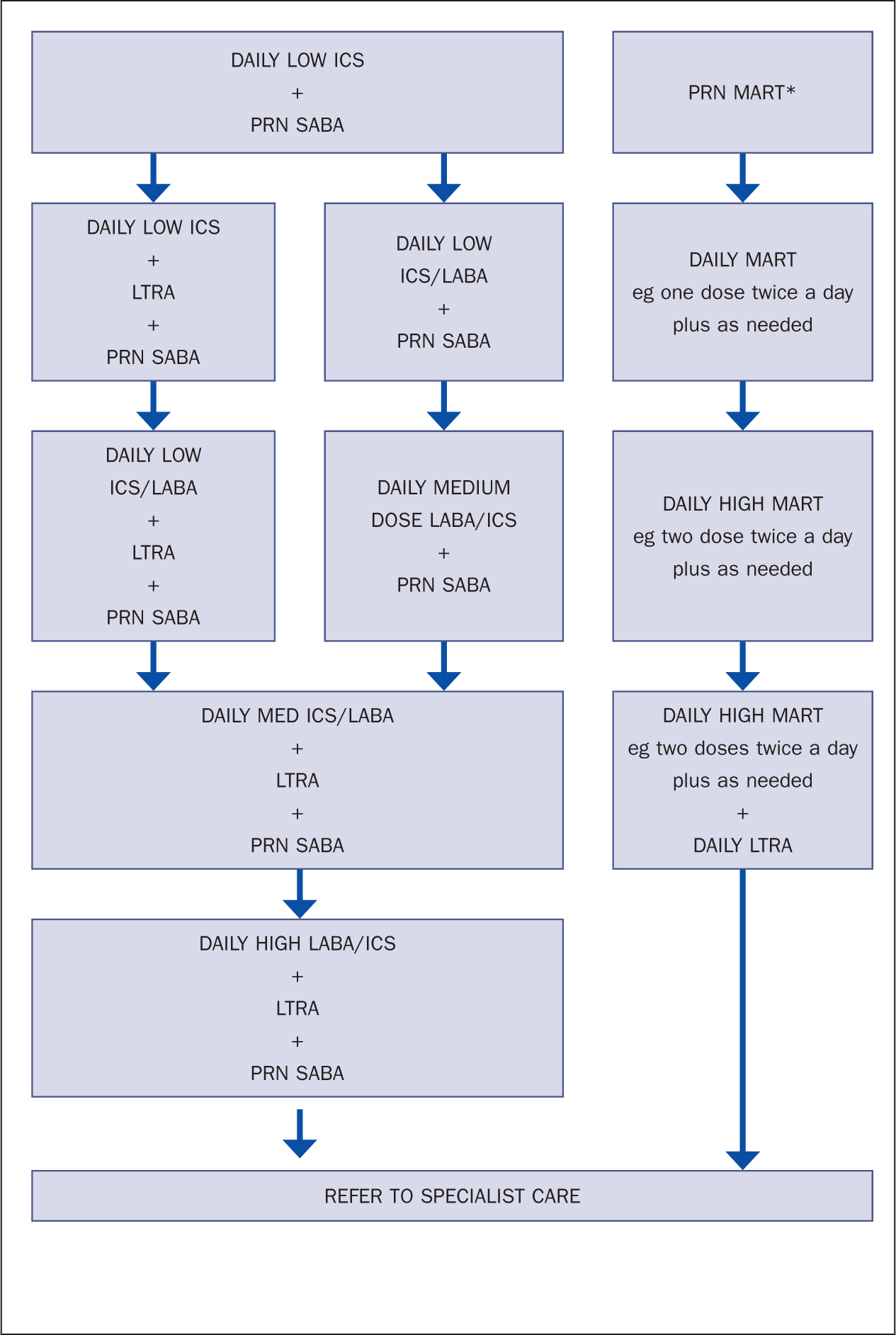 Figure 2. Examples of adult asthma pharmacological pathways, based on composites of NICE (2017), BTS/SIGN (2019) and GINA (2020) guidance
Figure 2. Examples of adult asthma pharmacological pathways, based on composites of NICE (2017), BTS/SIGN (2019) and GINA (2020) guidance
Inhaled corticosteroid
Initial therapy as recommended by BTS/SIGN (2019) and GINA (2019) should consist of low-dose ICS daily, plus as-required SABA. The NICE (2017) guideline suggests that SABA alone (without ICS) can be used initially. However, this guidance lags two years behind BTS and GINA.
ICS prescribing examples:
- Children: Clenil 50 mcg MDI, two puffs twice a day with Volumatic spacer
- Adults (DPI): budesonide Easyhaler 200mcg, two puffs twice a day
- Adults (MDI): Qvar Easy-breathe 100 mcg, one puff twice a day.
Short-acting B2 agonist
Prescribed as-required, SABA therapy is usually given as an adjunct to regular preventer therapy.
SABA prescribing examples:
- Children: Salbutamol MDI 100mcg with AeroChamber Plus with medium mask, child 1–5 years old, 1 to 2 doses as needed
- Adult (DPI): salbutamol Easyhaler 100 mcg, two doses as needed
- Adult (MDI): salbutamol Easy-breathe 100 mcg, two doses as needed.
Long-acting B2 agonist
Addition of LABA can be considered for those patients who are not controlled on ICS, as adding LABA to ICS is clinically more effective than doubling the dose of ICS (Kankaanranta et al, 2004). LABA should be prescribed with ICS in a combination inhaler, to avoid it being taken without ICS (BTS/SIGN, 2019). Given in combination in the same inhaler as ICS, LABA therapy can be used as a traditional once or twice daily preventer, or as maintenance and reliever therapy (MART).
Some LABA/ICS combinations are fixed-dose and others are adjustable. Adjustable dosing enables patients to increase and decrease their therapy in line with their symptoms and/or peak flow. Some patients may prefer a fixed-dose preventer, or a once-a-day treatment rather than twice-a-day treatment. Although it may seem logical that once a day would be preferable to twice a day treatment, there is no evidence to say one is any better than the other (Syed, 2015; Dwan et al, 2016).
Fixed dose LABA/ICS prescribing examples:
- Child: Symbicort 100/6, 1 to 2 doses twice a day
- Adult (DPI): Relvar Ellipta 92/22, one dose once a day
- Adult (MDI): Fostair MDI 100/6, 1 to 2 doses twice a day (preferably with spacer).
Maintenance and reliever therapy
When using a formoterol-based LABA/ICS combination, consideration can be given to using this as both a preventer and a reliever, known as MART, which takes advantage both of formoterol's rapid speed of onset, and its wide therapeutic range.
MART reduces the number of oral steroids, Accident and Emergency department attendances and hospital admissions when compared to fixed-dose combinations (Cates and Lasserson, 2009; Edwards et al 2010; Cates and Karner, 2013; Kew et al, 2013). The exacerbation rate is also reduced by up to 48% (Rabe et al, 2006) and patients take less medication, making MART a more efficient way of delivering inhaled therapy. Symbicort®, Duoresp®, Fobimux® and Fostair®(beclometasone/formoterol) are licensed for MART (see Table 1 for strengths and frequency of dosing), which involves taking a daily maintenance dose, plus extra doses as-required (rather than SABA). MART also has the added advantage of having only one prescription cost. The maximum number of inhalations can vary, depending on the ICS used.
Table 1. Comparison of licensed indications, published randomised controlled trials (RCT), systematic reviews, and meta-analyses of ICS/LABA combinations licensed for MART
| Drug | Device | Max doses/day | Frequency | Lower age | Published RCTs | Systematic reviews/meta analysis |
|---|---|---|---|---|---|---|
| Budesonide/formoterol MART | ||||||
| Symbicort 100/6 | Turbohaler | 12 for limited period | od or bd | 12 | 16 500 patients 7 studies over 5.5 years | 4 |
| Symbicort 200/6 | Turbohaler | 12 for limited period | od or bd | 12 | ||
| DuoResp 160/4.5 | Spiromax | 12 (for limited period) | od or bd | 18 | 0 | 0 |
| Fobumix 80/4.5 | Easyhaler | 12 (for limited period) | od or bd | 18 | 0 | 0 |
| Fobumix 160/4.5 | Easyhaler | 12 (for limited period) | od or bd | 18 | ||
| Beclometasone/formoterol MART | ||||||
| Fostair 100/6 | MDI | 8 | od | 18 | 1701 patients/1 year 56 patients/7 days | 0 |
| Fostair 100/6 | Nexthaler | 8 | od | 18 | 56 patients/21 days | |
Budesonide/formoterol MART can be given up to 12 inhalations per day (but for short periods only, with recommendation to seek health professional advice), is licensed from 12 years, and can also be prescribed once or twice a day. Beclometasone/formoterol MART can be given up to eight inhalations per day, and is licensed from 18 years. All come in DPIs, with the exception of Fostair®, which has both DPI and MDI, and is prescribed most often (Figure 3). There are no direct head-to-head studies comparing these treatments, therefore choosing which to prescribe may be down to device preference, or depth of evidence available. A search of the Cinahl database, plus data requests from AstraZeneca, Chiesi, Teva, and Orion Pharma showed that the majority of evidence about MART therapy has been conducted using formoterol and budesonide in a Turbohaler® (Table 1).
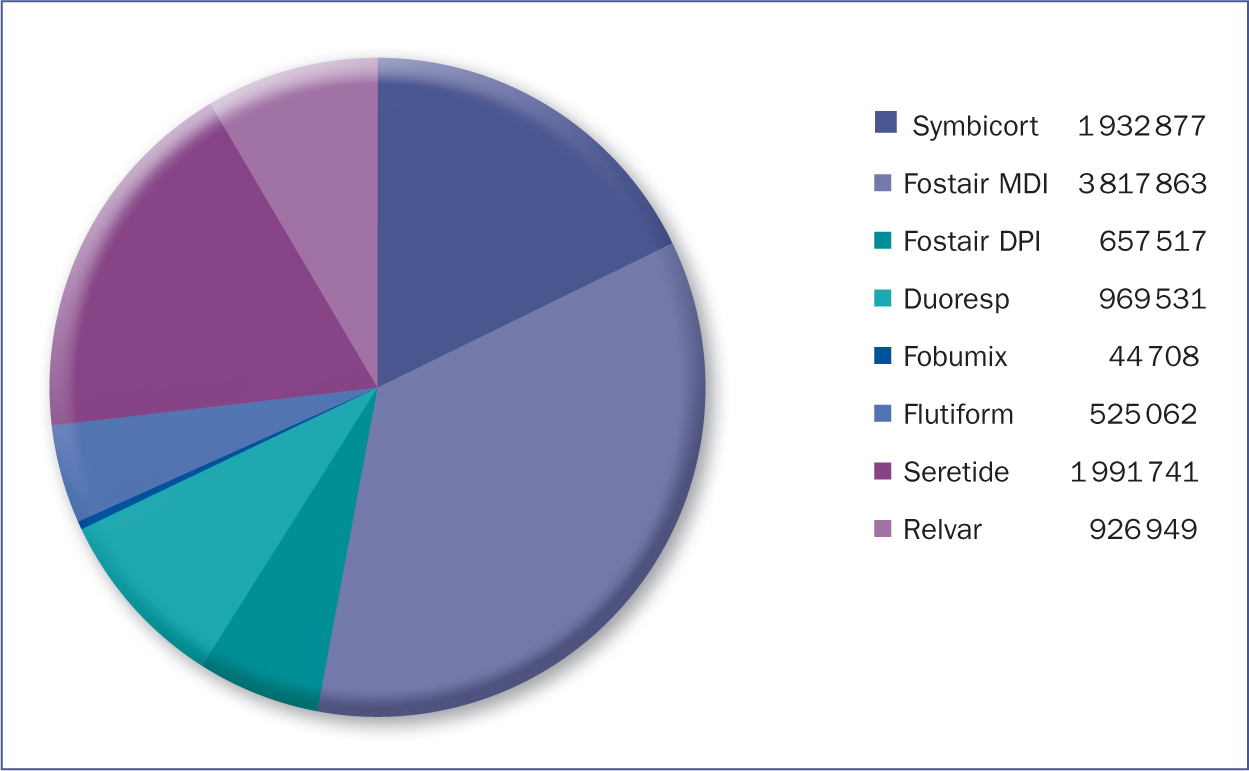 Figure 3. Number of prescriptions for ICS/LABA combination in England 2019.
Figure 3. Number of prescriptions for ICS/LABA combination in England 2019.
Some advice suggests that people taking MART therapy should have a ‘rescue’ salbutamol MDI and spacer (Keeley and Partridge, 2019). This would appear to be a logical and safe approach to any inhaled regime, not just limited to MART. Conversely, there is evidence that Symbicort Turbohaler® is as effective as an MDI and spacer during an asthma attack (Balanag et al, 2006).
GINA (2020) have reported further clinical trials of budesonide/formoterol combination, which have been studied with positive outcomes as a first-stage preventer- and-reliever, rather than regular ICS. These studies showed that ‘as-required’ budesonide/formoterol has significant therapeutic benefit in mild asthma (Bateman et al, 2018; Beasley et al, 2019; O'Byrne et al, 2018).
MART LABA/ICS prescribing examples
- Child 12 years old: Symbicort Turbohaler 200/6, 1 dose twice a day, plus extra as needed (max 12 doses per day)
- Adult: Symbicort Turbohaler 200/6, 2 doses once a day, plus extra as needed (max 12 doses per day)
- Adult: Fostair MDI 100/6, 1 to 2 doses twice a day, plus extra as needed (max eight doses per day).
Leukotriene receptor antagonists
At the next step-up in asthma treatment, BTS/SIGN (2019) recommend either increasing the daily dose of ICS or the addition of an LTRA. Conversely, NICE (2017) have recommended the use of LTRA therapy before the addition of LABA, with the rationale that the cost of LTRAs are slightly lower, and only slightly less clinically effective. Some people may prefer to take a tablet once-daily, rather than increasing their inhaled therapy. Guidance for children under 5 years old suggests that LTRA may be given before ICS (BTS/SIGN, 2019).
LTRA prescribing examples (medicines.org.uk, 2016):
- Children 6 months to 5 years: montelukast 4 mg oral granules, 1 sachet once a day in the evening.
- Children 6 to 14 years: montelukast 5 mg chewable tablets, 1 tablet a day taken in the evening
- Adults (over 15 years): montolukast 10 mg once a day in the evening.
Specialist care
If LABA/ICS plus LTRA are failing to maintain control, then strong consideration must be given to referral to specialist care (NICE 2017, BTS/SIGN, 2019).
Long-acting muscarinic antagonists
Tiotropium via the Respimat® is currently the only LAMA to be licensed for asthma. However, the evidence discussed in three systematic reviews shows that while there are some benefits to adding LAMA therapy to existing ICS/LABA, these benefits were not entirely clear (Evans et al, 2015, Andreson et al, 2015; Kew et al, 2016). It is perhaps for this reason that BTS/SIGN (2019) have classified LAMA as a ‘specialist therapy’ only.
Oral steroids
For treatment of an asthma attack or exacerbation, the use of short courses of oral prednisolone can rapidly reduce inflammation and gain control. Some patients can be equipped with a stand-by or rescue pack of oral steroids to keep in case of attack, which should be undertaken as part of education and self-management, so that the patient knows how to recognise a worsening condition, when to commence their rescue oral steroids and that they should contact their GP immediately. Continuous or regular use of oral steroids should not be commenced in primary care. Some patients with very severe asthma may benefit from initiation and monitoring by specialist asthma services. The risk of side effects such as osteoporosis, diabetes, and hyperlipidaemia are not insignificant.
Conclusion
ICS remain the mainstay and most important part of asthma treatment, as they prevent symptoms and inflammation and have slow but positive and long-lasting effects on the airways. ICS are safe, well-tolerated, and are clinically effective. Short-acting B2 agonist bronchodilators provide rapid symptom relief, while longer acting bronchodilators provide excellent add-on therapy. The role of leukotriene receptor antagonists have changed in recent years and can be used before the addition of LABA. LABA therapy, when added to ICS in a combination inhaler, has been shown to be safe and effective when used either as a fixed-dose or adjustable maintenance dose, or with greater clinical efficacy when prescribed as MART. In addition, future use of formoterol-based ICS combinations could be used ‘as required’ as firstline initial therapy for mild asthma.
KEY POINTS:
- Inhaled corticosteroids are the most important element of asthma therapy
- Bronchodilators play a significant role as an adjunct to inhaled corticosteroids
- Long-acting B2 agonists can be used in combination with inhaled corticosteroids to increase clinical effectiveness
- Leukotriene receptor antagonists can be used instead of or as well as long-acting B2 agonists
- There is a large body of evidence supporting the use of ICS/LABA therapy, especially as maintenance and reliever therapy.

